Science:
Subtopics

Victoria's new state fossil emblem: Koolasuchus cleelandi
After 11,563 votes, the 125-million-year-old monster amphibian has been confirmed as Victoria’s fossil emblem.

Jurassic relict: a new family of Brittle Stars
Long-armed and bristling with teeth, this Brittle Star is a marine relict.

Resurrected: native Australian mouse back from extinction
Once lost, now found.

Climate change and the environment program
Be a part of something bigger in 2021. Generating conversations about climate change, the environment, science and our society.

Letter from a Tasmanian Tiger
Ever wonder about the life of a museum specimen?

Bats: the flying mammals in dire need of a PR manager
Separating fact from folklore

The trials and triumphs of a trailblazing scientist
Hope Macpherson was a woman who broke barriers.

The platypus: a unique and vulnerable Australian
What can we do to help the platypus?

Nemo found: new species of dancing peacock spider named
Meet the newest dancing peacock spider, Maratus nemo.

Catching Lightning
A fulgurite that took microseconds to form, a moment to break and over 50 years to reassemble.

Investigating the IOT: deep-sea science
A journey into Australia's unexplored deep-sea Indian Ocean Territories.

A Lord of the Rings-like marine landscape revealed in Australia’s Christmas Island Territory
Take a dive into a Lord of the Rings-inspired seascape.

Our addiction to plastic
Lifting the lid on plastic: the good, the bad and the ugly.

Under fire: The animals threatened by Australia’s bushfire crisis
If there’s one place in the world that knows about extinction, it’s a natural history museum.

The puzzle of Melbourne’s small and ancient seals
‘Landmark’ study rewrites the history of seals in southern Australia thanks to fragmentary fossils washed up on a Melbourne beach.

Eleven incredible meteorites
The Murchison is just one of many strange and storied space rocks held by Museums Victoria. Here are 10 others—as well as one which disappeared.

Backyard ballistics: Australia's first DIY satellite
A DIY guide to reaching outer space.

Wedderburn meteorite
A meteorite from the core of another planet contains one of the rarest minerals on earth.

Skynotes
Our monthly newsletter on where to find the brightest planets in Melbourne.

Unboxing the museum: Deinocheirus claws
The racks and collections in the museum stores contain many objects and memories. This is Collection Manager at Museums Victoria, Tim Ziegler's personal story of just one.

Who wielded the Otway Claw?
Meet Victoria’s top Cretaceous predator—a dinosaur that would have eaten Velociraptor for breakfast.

Who are the moth hunters?
Meet the citizen scientists who are, literally, discovering new species in their backyards.

What good are spiders?
Apart from their intrinsic right to be here, spiders do humans a power of good as well.

Eight myths about snakes
Some common misconceptions, and what you can do to keep snakes out of your yard.

The first taipan to be milked for venom
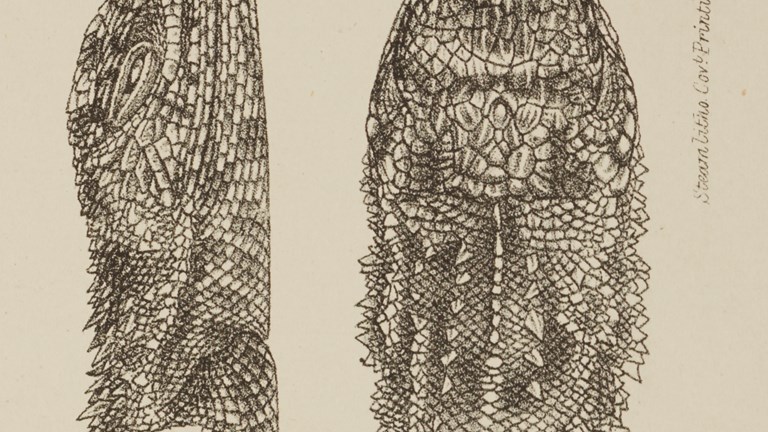
When is a creature extinct?
And does this dragon still exist?

Listening for Nature
Researchers are using advanced digital tools to listen in on animal sounds and map who is where from croaks and calls.

The colour of birds' eggs
The aesthetic qualities of a bird egg can tell you a lot about the species that laid it.

A.J. Campbell on Google Arts & Culture
Passionate ornithologist, naturalist and photographer.

The first donation to the Museum
Mounted specimen of the Little Pied Cormorant

Why the 2010s were the decade of the peacock spider
How disruptors and digital technology led to a frenzy of scientific discovery.
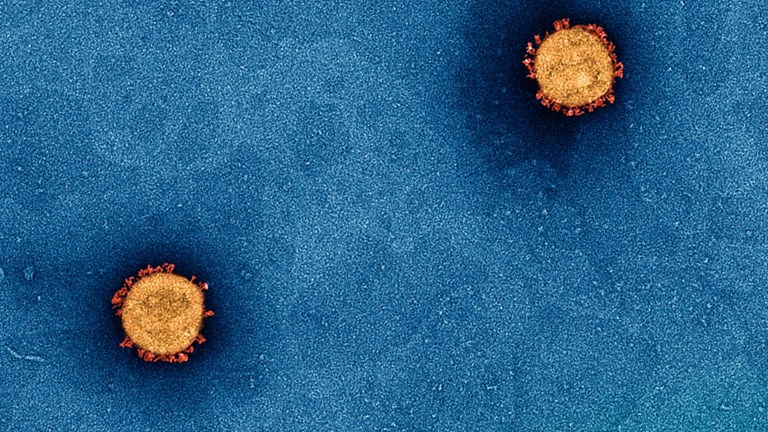
Vaccines: saving millions one dose at a time
How the history of vaccine development shaped our ability to respond to worldwide pandemics.

The Art of Science
Museum Victoria's archive of artworks, working drawings and rare books traces the development of scientific art and provides a glimpse into a world of uncommon beauty.

Forest Secrets
At the heart of the museum is a piece of Victoria's mountain landscape with tall eucalypts, ferns, rare plants and wildlife. Delve in to the secrets of our Forest Galley.

600 Million Years: Victoria Evolves
How did life on Earth come to be the way it is and what happened in our part of the world?

Dinosaur Walk
The Dinosaur Walk exhibition brings dinosaurs, pterosaurs and megafauna to life. Learn about the 17 skeletons of prehistoric animals on display!

Dynamic Earth
Uncover the story of our planet. Dynamic Earth is a stunning exhibition that uses thousands of minerals to tell the story of our ever-changing planet.

Discover Documentary: Taxidermy
Museums Victoria owns some of the world's most iconic taxidermy specimens from the famous Phar Lap to the infamous Sad Otter. There are over 16 million items in our collection and over 6 thousand of these are taxidermy. But why do scientists still use physical specimens?

Discover Documentary: Bees
In this episode of Discover, Dr. Ken Walker takes us into Museums Victoria's Entomology Collection. As an expert in native bees, he explains how these tiny creatures have a major global impact.

Discover Documentary: Rats
The Indonesian island of Sulawesi is home to a unique mix of Australian and Asian animals found nowhere else on Earth, including a number of rat species that are one of a kind. Find out how research being conducted by Dr Kevin Rowe and his colleagues into these rats is helping us to understand the evolution of life on our planet.

Discover Documentary: Deep Sea
Scientists are only just starting to uncover what lives in the deepest parts of the world's ocean. Find out about some of the weirdest and most wonderful with Dr Martin Gomon, Curator of Ichthyology and Melanie MacKenzie, Collection Manager of Marine Invertebrates.

Discover Documentary: Prehistoric marine mammals
What creatures swam in Australia's oceans millions of years ago? In this episode of Discover, palaeontologists Dr Erich Fitzgerald and Tim Ziegler explain how they find and prepare the fossils which are revealing the secrets of Victoria's prehistoric marine mammals.
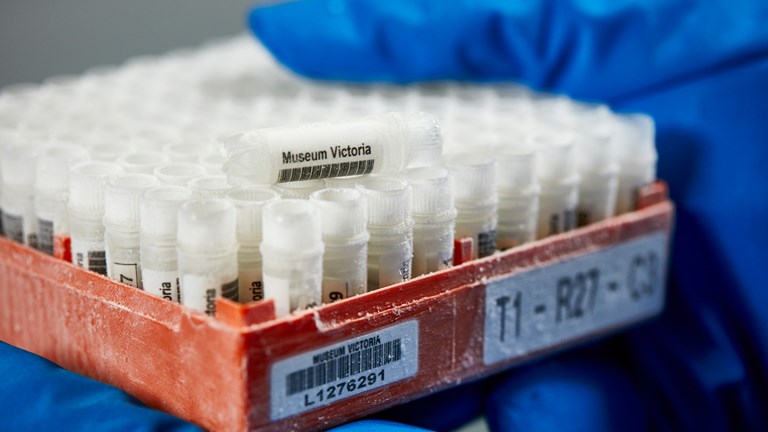
Discover Documentary: Biobank
Discover the Ian Potter Australian Wildlife Biobank: a state-of-the-art liquid nitrogen storage facility containing tissue, venom, and even live cells.

Discover Documentary: The Murray Explored Bioscan
Following in the footsteps of William Blandowski, the Museum's first curator.

Discover Documentary: Conservation
From wedding dresses to meteorites, our Conservation team protect and preserve over 15 million objects in Museums Victoria's collection. This episode of Discover takes a look at how they do it!

Planet or dwarf planet?
A group of astronomers are trying to reclassify Pluto as full 'planet'.

Melbourne's annual congregation of Giant Spider Crabs
Every year, thousands of Giant Spider Crabs congregate in Port Phillip Bay ahead of their annual winter moult.

Australia's megafauna
Australia’s megafauna were unique, and included giant marsupials, huge flightless birds and giant reptiles.

The Murchison meteorite
The story behind one of the most studied meteorites, which fell to earth near Victoria in 1969.

How do diamonds get their colours?
And what's so special about the pink ones?

Blue-ringed Octopus
Small but deadly, this animal runs rings around the competition.

Creature of the deep
Its evocative appearance is not the only thing that makes this deep sea creature fascinating and important.

What are these swarming beetles in my garden?
Each January, the Museum receives many enquiries about swarms of beetles in suburban gardens in and around Melbourne

Predator vs predator
Wasps give huntsman spiders a taste of their own medicine.

The science of poo
Poo is truly fascinating stuff.

Do centipedes really have 100 legs?
Counting the feet on these ancient invertebrates.

Victoria's nudibranchs
The butterflies of the sea.

Moon rock on display
Museums make it possible to see specimens from faraway places that you won't get the chance to visit yourself.

Buzz off!
How do you study a bee so small it can barely be seen?
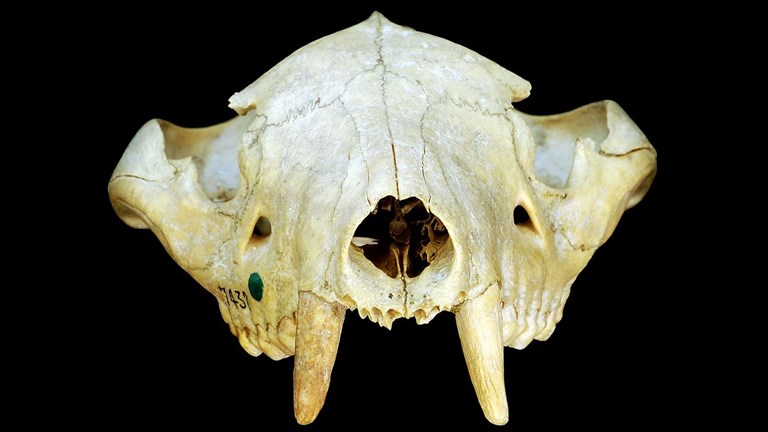
Small(er) is beautiful
Megafaunal giants…in miniature

Cephalopod Catalogue
Third and final volume by MV scientists now available

Outreach koala
Preparing a specimen for education programs.

Bountiful Mallee
Close encounters with Mallee insects.

Whale vs shark
Evidence of shark bite 24 million years ago.

Sea anemone feast
Ever bitten off more than you can chew?

Our first Director's vision for a University Botanical Garden
How Melbourne University's System Garden came to be.

Ancient whales had more bite than today’s gentle giants
The cutting edge of whale evolution.

Northern exposure: fossils of a southern whale found for the first time in the north
A totally unexpected discovery.

Dream Deep
The story behind Joe Meldrum's improvised dance piece in the infinity room of Scienceworks' LightTime exhibition.

Secrets from beyond extinction: the Tasmanian tiger
The entire thylacine genome has now been sequenced, revealing the apex marsupial predator was in poor genetic health and may have struggled to fight disease had it survived.

A good possum is hard to find
The story of Leadbeater's Possum is a remarkable one.

The Larsen-C Benthos expedition
What lurks beneath the Antarctic ice? Museums Victoria's Mel Mackenzie is one of the lucky scientists on a voyage south to find out.

Putting names to faceless fishes from the abyss
Understanding which fish species occur where, and discovering new fish species, is the starting point to managing marine biodiversity.

Extinct Tasmanian Tiger now back in 3D
Using 3D scanning, researchers are peeking under the preserved skin of Tasmanian tiger specimens to reconstruct its growth and development.

A treasure trove of freshwater fish biodiversity
The Kimberley region in Australia’s northwest is one our last great pristine unspoilt places, and a hotspot for species discovery.

Augmented Humans
Melbourne is home to exciting new innovations combining medicine, design and technology. Many of these have the power to augment human abilities.

Eulogy for a seastar, Australia’s first recorded marine extinction
Today, I am writing a eulogy to the Derwent River Seastar (or starfish), that formerly inhabited the shores near the Tasman Bridge in Hobart, Tasmania.

Butterflies of the night
The aim of the "George Lyell Collection: Australian entomology past and present" project is to examine the George Lyell collection scientifically and culturally, and to share discoveries with the wider community.

Pressed Orchids
Unlike their Asian counterparts, Australian orchids like the rosy spider orchid, pictured below, are beautifully understated. Delicate and beautiful, they are not flashy like the orchids available at your local nursery.

The sting of the final letter
Transcribing George Lyell's final documents.

Light sheets
Peter Marriott talks moth bioscans in the Otways for National Science Week at Melbourne Museum.

Kindred spirits
Between 1891 and 1947, George Lyell and Gustavus Athol Waterhouse's regular correspondence shared a passion for moths and butterflies.

The 700
More than 700 individuals whose combined efforts, starting in 1984, have brought to light fossils in what has been dubbed by one of them “The Dinosaur Dreaming Project”.

Moths are beautiful too
At October's Nocturnal event, a multidisciplinary team of Museum workers and a guest speaker from University of Melbourne got together to present items from the George Lyell Collection to visitors.

On the wing
Professor Deirdre Coleman, Nik McGrath and Simon Hinkley join Richelle Hunt in the ABC Radio Melbourne Studio.

George Lyell's letters
In addition to the development of his moth and butterfly collection, George Lyell’s letters reveal much about his personal relationships.

What is the most venomous animal in the world?
And how its deadly venom could save lives.
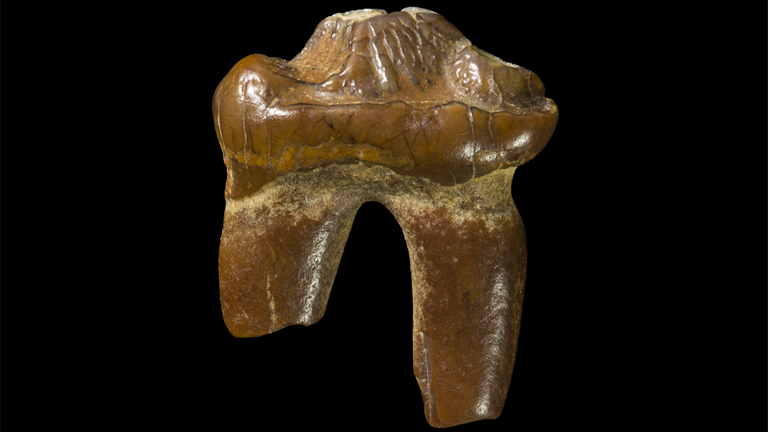
Rare fossil tooth find sheds a light on Australia’s distant past

Who’s digging in my lawn?
Learn about land crayfish and their burrows.
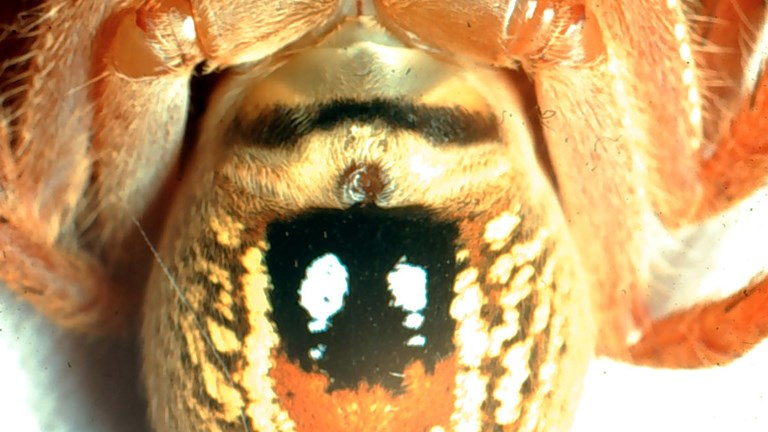
Is my spider a boy or a girl?
At some times in a spider’s life it is easy to tell, but at other times it can be impossible.

Scorpion facts and fallacies
Are all scorpions dangerous? Answers to this and other common scorpion questions.

Birds and birdwatching
Birds are highly visible and vocal, spectacularly diverse and fascinating to watch and study.

Your eight-legged housemates
At Museums Victoria we are often asked: “What kind of spider is this? It was in my house! Is it dangerous?”

Nowhere to hide?
Searching for the elusive Tooarrana in the wake of the fires.

Colour Variations: It doesn't look like that in the book

Astronomy and the art of scientific storytelling
Capturing the awesome story of the universe.

Museums, Specimens and Taxidermy
Bringing the dead to life - sort of!

Tasmanian Tiger: lessons from the last of its kind
Museum specimens are enabling new discoveries about extinct species, long after they are gone.

Wild goodbyes
We've been blown away by the number of people who have reached out to us about Wild—here are some answers to your most often asked questions.

Anne Bermingham, a scientific pioneer of radiocarbon dating
Meet the woman who started Australia’s first radiocarbon dating lab.

Sylvia Whincup, a prolific collector and groundbreaking mineralogist
Sylvia amassed a collection of thousands of specimens and 167 new species.

The doyenne of dinosaur discovery
Melissa Lowery has a rare talent for finding dinosaurs.

Flight of fashion: when feathers were worth twice their weight in gold
The high price of feathered hats in the 1800s.
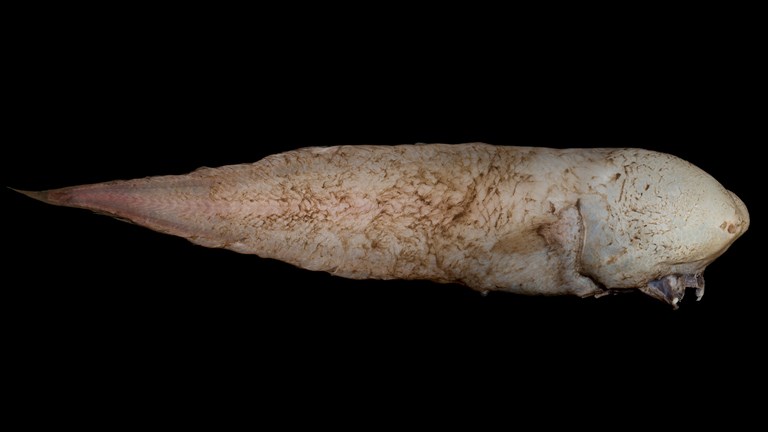
Faceless Fish and the deep-sea voyages that found it
Take a dive into the deep sea with the Faceless Fish.

Native and introduced rats: some quick and dirty facts
Can you tell a native rat from a pest species?

Glowing animals: understanding bioluminescence and biofluorescence
What do a Platypus, a Dragonfish and a Scorpion all have in common?

A Japanese Devilray, far from home
Collecting the first Victorian record of the Japanese Devilray.

10 really big things in the Museums Victoria State Collection
With millions of objects to choose from, what are the some of the biggest things?

Unboxing the museum: Deinocheirus claws
The racks and collections in Museum stores contain many objects and memories this is Tim's personal story of just one.

Why are there so many whale skeletons in museums?
Whales grace the halls of many a natural history museum, but they are there for more than just show.

New species of squat lobster helping to revive an Aboriginal language
How the naming of a new species of squat lobster has helped an Aboriginal community reconnect to its ‘sleeping’ language.

Birthday honours: 21 of our favourite things about the Melbourne Museum
Fascinating facts about Melbourne’s favourite museum.

How do deep-sea creatures survive in the crushing dark?
Meet some of the weird and wonderful animals that have adapted to life in the deep sea.

Top spider myths
A myth for each leg: from the deadly Daddy Long-legs to swallowing spiders in your sleep.

Recovering a rarity: what happens when a dead Fin Whale washes ashore?
The stranding of a dead whale offers scientists a rare opportunity to study these elusive giants.

The chance discovery of one of Victoria’s oldest fossil plants, 400 million years in the making
A series of unlikely events that led to the discovery of a 400-million-year-old fossil, shedding new light on the evolution of Australian plants.

More Australian species added to the endangered list
Without action, museum collections may be the only evidence some of these endangered species existed.

If books could kill: poison, heavy metal and literature
Poisonous books are a legacy of fashion and industrial practices that prioritised beauty above all else. And the heavy metals left behind are still causing headaches for libraries and museums to this day.

Lunar New Year: 10 Australian tigers
Think you know tigers? What about those found in Australia?

Jan Watson: diving into the unknown
One of Australia’s first scientific divers, Dr Jan Watson used her tenacity, skill, and passion to realise her underwater ambitions.

Isabel Cookson: one tough ‘Cookie’
One of Australia’s first professional female scientists, Isabel Cookson was a ground-breaking palaeobotanist.

Forbidden objects in the museum's collection
What do a doll, religious text, gun, and surprised-looking cat have in common?

Rhyll Plant: sowing the seeds of scientific art
Half a century as a scientific artist has taken Rhyll Plant to some unexpected places, but where did it all begin?

Know your bones: what is a ‘real’ fossil?
Fossils are the only means we have to study and display ancient animals, but what makes a fossil ‘real’?

The mighty Muirfield Seamount
From ship slayer to protected wonderland, the Muirfield Seamount has a long history of surprising seafarers.

This small rock holds the story of Antarctic exploration before the Heroic Age
Discover the rock that unlocked one of the biggest mysteries of Antarctica and the incredible story of Carsten Borchgrevink, the explorer determined to get there first.

Lunar New Year
This Lunar New Year, come for a tour of the Chinese Zodiac through the Museums Victoria collection.
How poo shapes a city (and other scatalogical stories)
What do Melbourne’s laneways, the Spotswood Pumping Station at Scienceworks, and termite mounds all have in common?

A glimpse into the past, to enlighten the future of biodiversity on Earth
What can we learn from the first scientific descriptions of animals and the Earth’s biodiversity, written hundreds of years ago?

Live bugs, in a museum?
What does the thought of picking up a bug do to you? It’s just a day in the life for our Live Exhibits keepers.

Reproduction can be a lonely task for a deep sea fish
What does it take to reproduce in the cold, dark and lonely world of the deep sea?

Deaccessioning: Why do museums remove objects from collections?
The public collections found in museums across the world are a historic record. But that does not mean everything stays there forever.

These pieces of atomic glass are the remnants of the first nuclear bombs
Forged in the fury of nuclear explosions, like Oppenheimer’s Trinity test and British bombs at Maralinga, atomic glass is more than a curio in museum collections.

What’s in a name? An animal’s can be misleading
When is a tiger not a cat, a fox not a canine, and a jelly fish not a fish? Unfortunately, more often than you might think.

125-million-year-old footprints rewrite the history of birds in Australia
Ancient bird tracks found on Australia’s southeast coast, could be the oldest evidence for avian dinosaurs in the Southern Hemisphere.

19-million-year-old fossil jaw bone hints the biggest whales first evolved somewhere unexpected
A newly described fossil from South Australia is making waves in our understanding of where and when whales evolved titanic body sizes.

How do we know wombats can run at 40km/h?
Uncovering the truth behind a wombat's top speed.

The Cape Paterson Claw Theropoda
This claw - the first dinosaur bone found in Victoria - is commonly known as "the Cape Paterson Claw".

Melbourne Observatory collection
Over 400 objects and images relating to the operation of Melbourne Observatory from 1863 to 1944.

Venoms & antivenoms at Museum Victoria
Through necessity, Australia is a world leader in venom and antivenom research.

Help us document biodiversity – record your own sightings

Wolf spiders
Some Wolf Spiders shelter in permanent burrows, others wander during the day.

Australia's oldest insect specimen
A Common Evening Brown collected in China in 1742.

Sudell's Frog
A burrowing species that usually come to the surface only after summer rains to breed.

Spiny Spider
Also known as the Christmas spider or jewel spider.

Eastern Banjo Frog
Burrows in the soil and are sometimes found when people dig in their backyard.

Redback Spider

Leaf Green River Tree Frog
These frogs frequent fast-flowing rivers and are excellent swimmers and jumpers.

Diprotodon tooth
A tooth fossil showing markings which appear to be man-made.

Spotted Grass Frog
A medium-sized frog with distinctive large regular-shaped brown or olive green blotches along its back.

Sampling the Abyss
The Sampling the Abyss voyage, 15 May–16 June 2017

Great Melbourne Telescope
Great Melbourne Telescope project website. The goal is to restore the telescope to working order so that it may be used for educational and public viewing.

Minerals and Gemstones of Victoria
See some of Museums Victoria's collection on Google Arts & Culture.

CSIRAC - the world's oldest intact first-generation electronic computer
When it was built CSIRAC was at the cutting edge of the new field of computing.